Packaging leak-tightness is often the last thing you think about… until the day a product leaks, gets contaminated, or fails quality control. In the cosmetic, pharmaceutical, food & beverage, and chemical packaging industries, ensuring packaging integrity is not an option: it is a regulatory, commercial, and safety requirement.
But faced with the diversity of equipment available on the market — vacuum testers, compression testers, immersion testers, internal pressure testers — how do you know which one truly matches your needs? This guide walks you through the process step by step to help you make the right choice.
Why Leak Testing is Essential
Before choosing equipment, it is worth recalling what we are trying to avoid. A defective seal on a food pouch can cause bacterial contamination. An improperly sealed pharmaceutical vial can compromise the sterility of a medication. A leaking cosmetic doypack on the shelf represents a direct loss of revenue and a reputational risk.
Leak testing makes it possible to detect these defects — often invisible to the naked eye — before the product leaves the factory. It is part of a broader quality control approach and compliance with international standards such as ASTM standards (D3078, F3039, D6653, D4991). An undetected defect in production can lead to costly batch recalls, regulatory penalties, or worse, a consumer incident. Implementing a rigorous leak testing process therefore means protecting product quality, brand reputation, and end-user safety all at once.
The Main Categories of Leak Testing
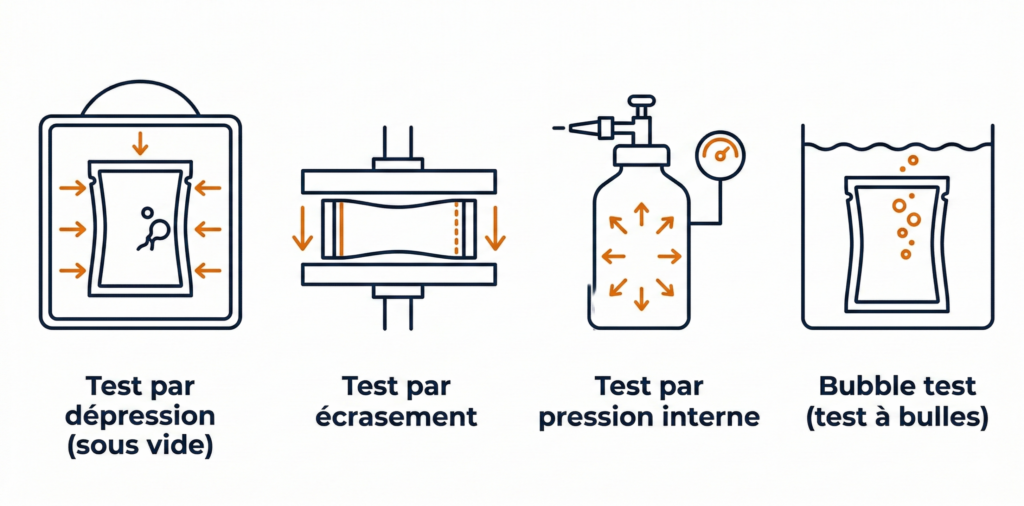
The vacuum test (or negative pressure test) consists of placing the packaging in a sealed chamber, then creating a vacuum. If the packaging has a leak, air or liquid escapes from it, revealing the defect. This is the most widely used method for flexible packaging (pouches, doypacks, bags) and is notably described in ASTM D3078 and ASTM F3039 standards.
The compression test specifically targets the seals of tubes, pouches, and sachets. The packaging is mechanically compressed under a precise and controlled force. If the seal gives way, the defect is immediately detected. This method is particularly well-suited for inline production, enabling fast and repeatable inspection.
The internal pressure test consists of injecting air inside the packaging and measuring whether the pressure holds over time. Any pressure drop signals a leak. This method is effective for rigid or semi-rigid packaging.
The bubble test is a variant of the vacuum test with water immersion: the packaging is submerged in a tank, a vacuum is applied, and any leaks are revealed in the form of bubbles. It is a visual, straightforward, and standardized method (ASTM D3078).
The altitude simulation test replicates air transport conditions by subjecting the packaging to prolonged low pressure, in accordance with ASTM D6653 standard. It is particularly useful for products shipped by air or stored at high altitude.
The 6 Key Criteria for Choosing Your Equipment
1. The Type of Packaging to Be Tested
This is the starting point of any decision-making process. A cosmetic tube, a sterile pouch, a glass bottle, and a food doypack are not tested in the same way. For flexible packaging (pouches, bags, doypacks), the vacuum test is generally the most suitable. For heat-sealed tubes, the compression test is the industry standard. For rigid packaging (glass bottles, aerosols), an internal pressure test or a vacuum test with an appropriate chamber will be more relevant. If you work with a wide variety of formats, it is better to opt for a versatile solution capable of adapting to multiple packaging types.
2. The Industry Sector and Its Requirements
Depending on your industry sector, the standards to comply with are not the same. The pharmaceutical industry is subject to strict requirements in terms of traceability and data integrity, notably FDA 21 CFR Part 11 regulation. The food & beverage industry favors fast methods that are compatible with a production environment. The medical sector requires compliance with ISO and ASTM standards. The cosmetics industry, for its part, often seeks a balance between reliability, speed, and ease of use. Before choosing equipment, list the standards you need to comply with: your tester should natively incorporate the corresponding protocols, without having to configure them manually for each test.
3. The Usage Environment: Laboratory or Production Line?
Laboratory equipment and production line equipment do not have the same constraints. In a laboratory setting, the priority is precision, flexibility, and the ability to perform a variety of tests on development samples. In a production environment, the priority is above all speed, robustness, ergonomics, and ease of use by non-specialized operators. Also consider the physical footprint of the machine, its power supply (electrical, compressed air, or both), and its resistance to hygiene conditions if you operate in a food & beverage or pharmaceutical environment. The IP protection rating of the device is a useful indicator to check: an IP 54 rating, for example, guarantees protection against water splashes and dust.
4. Traceability and Data Management
In many industries, performing a test is no longer enough: you need to keep proof of it. Traceability requirements mean that the equipment must be able to record each result, associate it with a batch number, an operator, a date, and export this data in a secure manner. Make sure your tester features user access rights, an audit trail, automatic data backup, and that it is compatible with your quality management software (SPC, ERP such as SAP). These features, long reserved for high-end equipment, are now accessible on modern devices designed for regulated industrial environments.
5. Flexibility and Modularity
Your packaging evolves, and so do your product lines. Equipment that is too specialized can quickly become obsolete if your production changes. Opt for modular solutions, with interchangeable accessories (platens, chambers of different sizes) and the ability to create customized test programs tailored to your internal protocols. Modularity is also an economic argument: rather than investing in several specialized devices, a flexible piece of equipment can cover all your needs, from R&D development testing to daily production quality checks.
6. After-Sales Service and Calibration
A leak tester is a measuring instrument. Like any measuring instrument, it must be regularly calibrated to ensure the reliability of its results. Make sure the manufacturer offers a recognized calibration certificate (ideally COFRAC or LNE), and that after-sales service is available promptly in the event of a breakdown or sensor drift. Equipment designed and manufactured in France generally offers a significant advantage on this point: response times are shorter, spare parts are more readily available, and French-speaking technicians are on hand to support you over the long term.
Common Mistakes to Avoid When Choosing a Leak Tester
Focusing solely on the purchase price is probably the most common mistake. A low-cost piece of equipment that is poorly suited to your needs, difficult to calibrate, or lacking connectivity with your management systems will end up costing you far more in the long run in terms of non-conformities, production losses, and after-sales interventions.
Overlooking the question of standards is another classic mistake. If your customer or quality certification requires testing in accordance with ASTM D3078, you must ensure that your equipment incorporates this protocol natively and reproducibly — not just approximately.
Underestimating traceability requirements is a common pitfall outside of the pharmaceutical sector. Yet, even in the food & beverage or cosmetics industries, quality audits are increasingly requiring documented evidence of the tests performed.
Choosing a machine that is too rigid for a constantly evolving production is also problematic. Always prefer equipment whose accessories and programs are configurable, to keep pace with the evolution of your product range.
ACRN’s Recommended Solutions
Drawing on several decades of experience in industrial packaging quality control, ACRN designs and manufactures leak testing equipment in France, tailored to the requirements of the most demanding industries. Here are the two flagship solutions recommended based on your needs.
The AgiLeak — The Next-Generation Vacuum Leak Tester
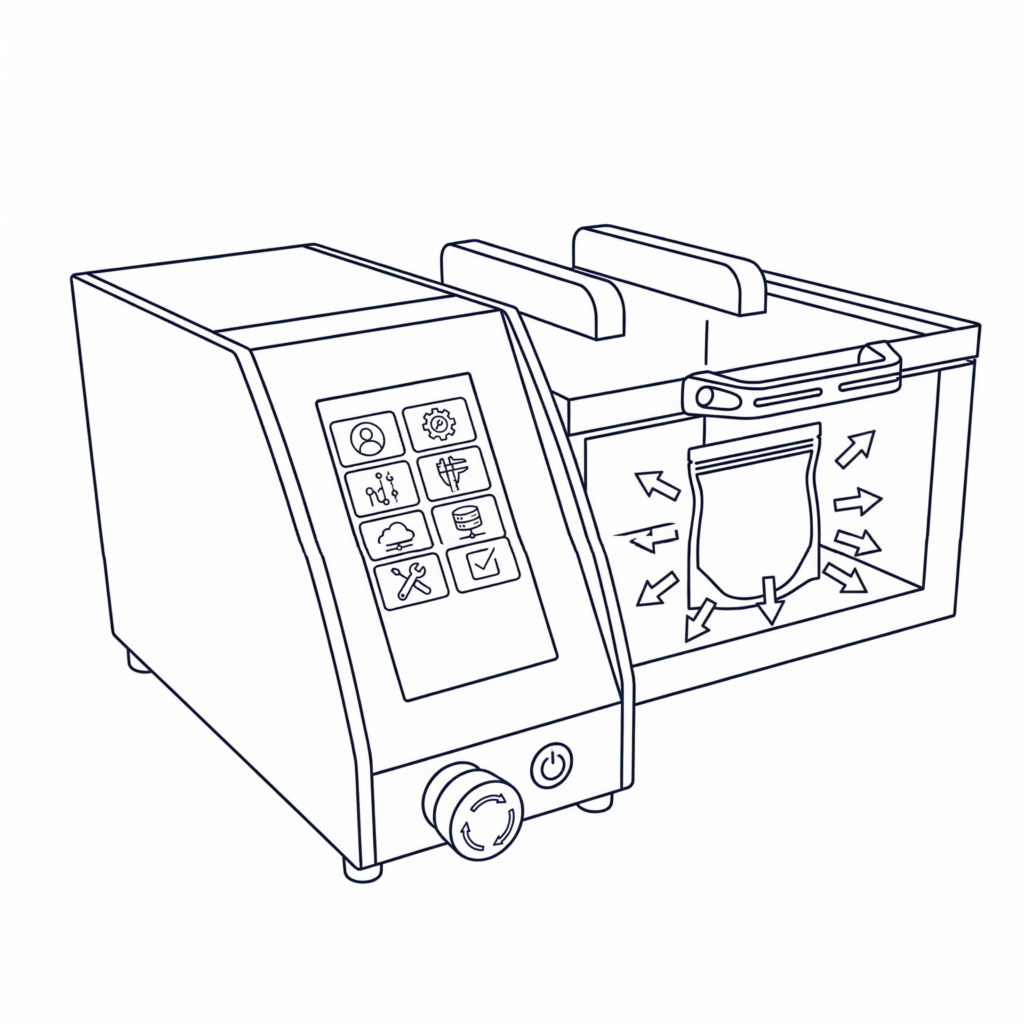
The AgiLeak is a vacuum leak tester designed to perform reliable, automated, and traceable tests on all types of packaging: flexible, semi-rigid, and rigid. Thanks to its intuitive 7-inch touchscreen, your operators can launch a test in under 5 minutes. It natively incorporates the protocols of ASTM D3078, F3039, D6653, and D4991 standards, and can perform a wide range of tests: leak detection, mechanical resistance, bubble test, air transport simulation, burst test, or fully customized programs. Available in 14 chamber sizes, it adapts to all your packaging formats. In terms of traceability, it meets FDA 21 CFR Part 11 requirements through its user access rights management, audit trail, and automatic data backup..
The PouchTube (ÉcraseTube) — The Seal Tester for Tubes and Pouches
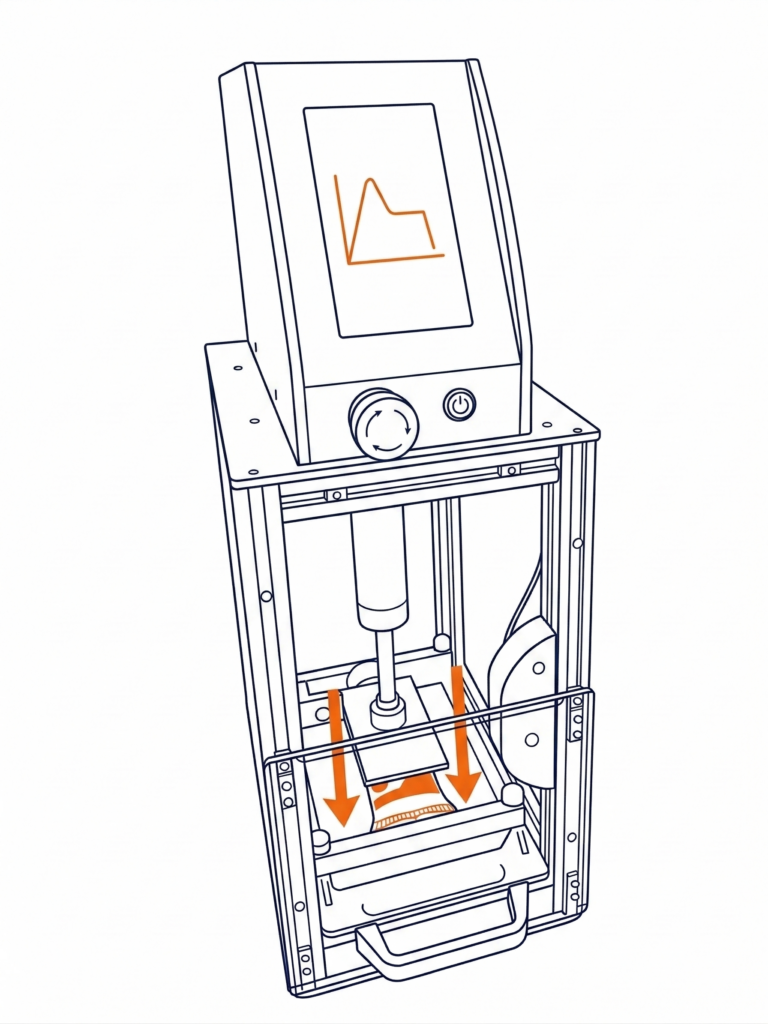
The PouchTube is the reference solution for checking seal integrity on tubes, pouches, and sachets, directly at the production line or in the laboratory. It can be used as a replacement for ATS tube seal testing devices. It offers a wide range of tests: compression resistance, cyclic testing simulating repeated opening and closing, internal pressure test, staircase test to determine compliance values, and custom tests according to your requirements. Its platens are interchangeable depending on the packaging to be tested, giving it maximum modularity. Delivered with an LNE/COFRAC calibration certificate, its results are exportable via USB or TCP/IP with SPC compatibility (Freeweigh, SAP). Maintenance-free, robust, and equipped with a 7-inch touchscreen with an IP 54 rating, it is perfectly suited for industrial production environments.
👉 Contact ACRN for Personalized Expert Advice